Recombinant lentivirus(LV) is an enveloped retrovirus with a single-stranded RNA genome, which is widely used for efficient gene delivery into mammalian cells. Smart’s current recombinant lentivectors are derived from human immunodeficiency virus-1 (HIV-1) and delivers high quality and high titer LV for overexpression, knockdown and knockout applications.

Fast turnaround

High purity, low endotoxin levels and minimal empty capsids for reliable performance

One stop solution for downstream stable cell line development
Stable cell line development could express exogenous genes constitutively, which is an important part for life science research and manufacture. At Smart, we recognize that having strong cell engineering capabilities is critical to support customer’s research. In order to provides the one stop solution from, our experts as well as accompanying technical expertise to help meet customers’ project needs.

One stop solution

Extensive experience - 13+ years

Project success rate - 98%
| Service Steps | Timeline | Description |
|---|---|---|
| Gene synthesis & cloning | 7-11 weeks | Plasmid contruction |
| LV packaging & purification | Cotransfection and packing cell line | |
| Titering detection | Concentration and purification Titering QC | |
| Stable cell line contruction | Virus infection and pressure screening | |
| Cell line detection | FACS detection of positive results for the cell line and passaging stability |
Note:
1. Please submit your request to the related BD.
2. The timeline is decided from sequence evaluation.
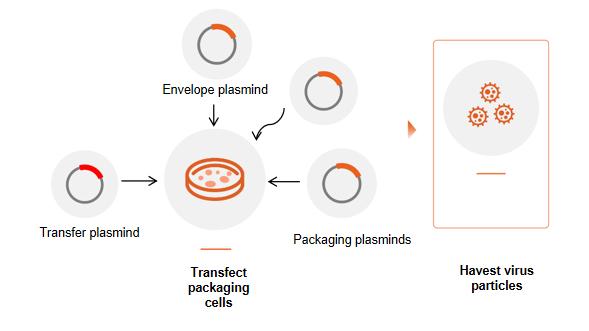
Co-transfect with transfer, packaging and helper plasminds for LV packaging, collect and puriy rAAV virus by PEG method. QC detectionby FACS for titer and infectivity determination.
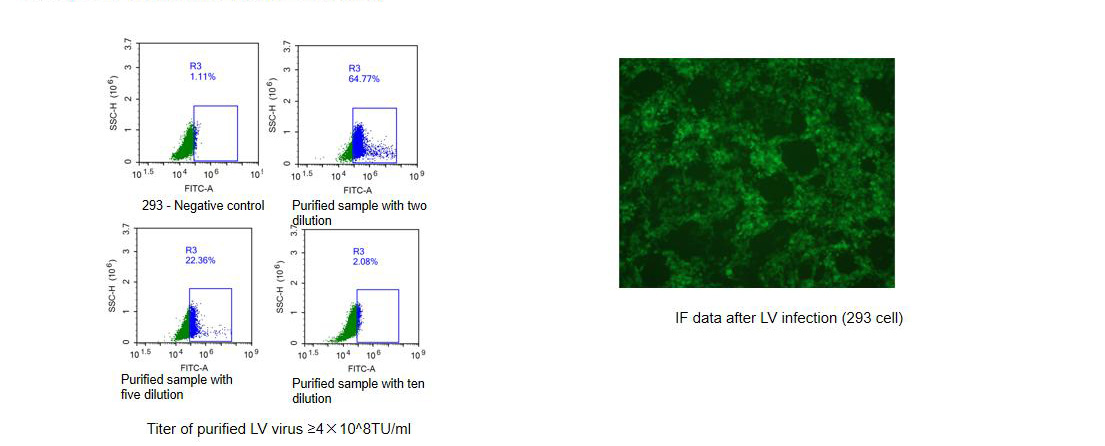
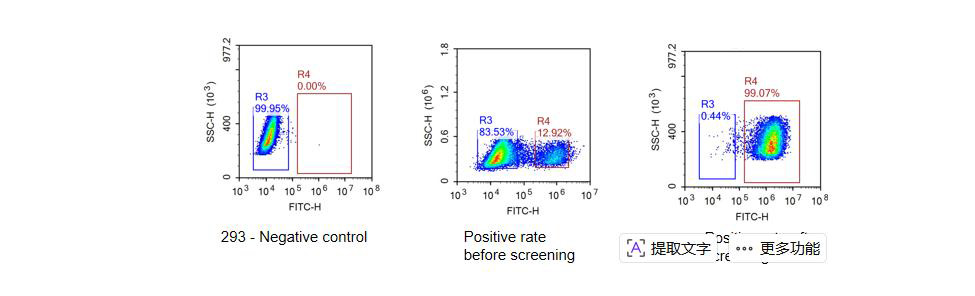
Constructing the stable cell line by using LV packing method to obtain the stable cell line with only two weeks and the positive rate more than 99%.
